  Energy dispersal also means that differences in temperature, pressure, and density even out. Roughly speaking, the second law says that in an isolated system, concentrated energy disperses over time, and consequently less concentrated energy is available to do useful work. For example, the second law implies that heat does not spontaneously flow from a cold material to a hot material, but it allows heat to flow from a hot material to a cold material. The second law of thermodynamics is an axiom of thermodynamics concerning heat, entropy, and the direction in which thermodynamic processes can occur. For any isolated system with a mass of more than a few picograms, the second law is true to within a few parts in a million. Such tiny systems are outside the domain of thermodynamics, but they can be investigated using statistical mechanics. But on scales of a few atoms, the second law does not apply for example, in a system of two molecules, it is possible for the slower-moving ("cold") molecule to transfer energy to the faster-moving ("hot") molecule. No violation of the second law of thermodynamics has ever been observed in a macroscopic system. Thermodynamics is a theory of macroscopic systems at equilibrium and therefore the second law applies only to macroscopic systems with well-defined temperatures. Thus, a heat engine with 100% efficiency is thermodynamically impossible. At least some of the energy must be passed on to heat a low-temperature energy sink. That is, it is impossible to extract energy by heat from a high-temperature energy source and then convert all of the energy into work. It is impossible to convert heat completely into work. For example, the electrical energy going into a refrigerator is converted to heat and goes out the back, representing a net increase in entropy.Ī third formulation of the second law, the heat engine formulation, by Lord Kelvin, is: This is allowable in a non-isolated system, however only if entropy is created elsewhere, such that the total entropy is constant or increasing, as required by the second law. Note that from the mathematical definition of entropy, a process in which heat flows from cold to hot has decreasing entropy. For example in a refrigerator, heat flows from cold to hot, but only when electrical energy is added. Informally, "Heat doesn't flow from cold to hot (without work input)", which is obviously true from everyday experience. Heat cannot spontaneously flow from a material at lower temperature to a material at higher temperature. If a system is at equilibrium, by definition no spontaneous processes occur, and therefore the system is at maximum entropy.Īlso due to Clausius is the simplest formulation of the second law, the heat formulation: (An exception to this rule is a reversible or "isentropic" process, such as frictionless adiabatic compression.) Processes that decrease total entropy of an isolated system do not occur. Thus, the system can either stay the same, or undergo some physical process that increases entropy. In an isolated system, a process can occur only if it increases the total entropy of the system. The formulation of the second law that refers to entropy directly is due to Rudolf Clausius: 
Thus, the theorems of thermodynamics can be proved using any form of the second law. There are many ways of stating the second law of thermodynamics, but all are equivalent in the sense that each form of the second law logically implies every other form (Fermi, 1936). 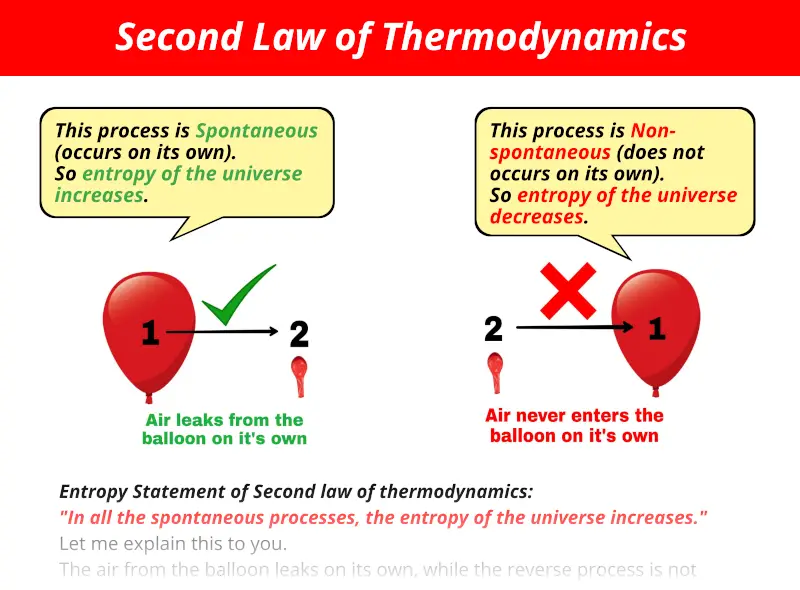
5.1 Perpetual motion of the second kind. #The second law of thermodynamics definition free#
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
